What if you could detect Alzheimer’s with a lens scan?
about
A simple eye scan to aid in the early diagnosis of a complex and devastating disease: Alzheimer’s.
Cognoptix is creating and developing a simple, innovative, non-invasive eye scanning test to aid in the early detection and diagnosis of Alzheimer’s Disease.
The key to effective preventive therapy and treatment of Alzheimer’s is the creation of an “early warning system” for the disease, before it progresses to the point where treatment options are limited. Cognoptix’s technology has demonstrated successful preclinical and clinical results as the company develops its diagnostic platform – which consists of a laser eye scanning device combined with an ophthalmic ointment which specifically identifies Alzheimer’s related proteins found in the lens of the eye.

"Cognoptix: Pioneering the Early Detection of Alzheimer’s Disease"
Cognoptix was recently profiled by MedTech Outlook as one of the Top 10 Non-Invasive Device Solution Providers for 2020.
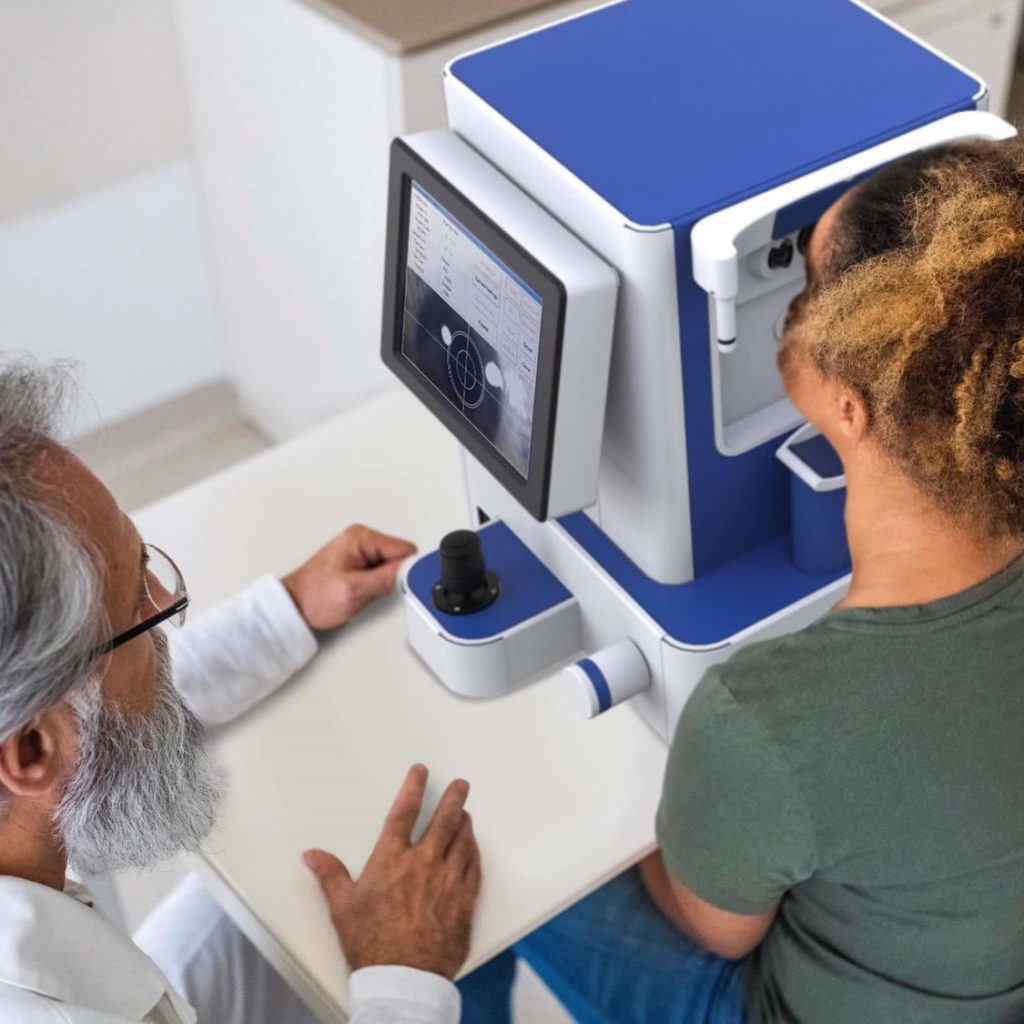
An Early Warning System to Enable Swift Intervention
Cognoptix’s goal is to provide clinicians with a highly accurate, reliable, inexpensive and widely available test that can be performed quickly by a general practitioner in an office setting, to enable swift intervention and ongoing monitoring of patients.
The company is interested in partnering with major pharmaceutical companies, licensing its laser eye scanning technology for use in the development of Alzheimer’s drugs, as an enabling technology for patient titration and monitoring in clinical trials.
Our Mission
To aid in the early detection, diagnosis and prognosis of Alzheimer’s disease through a simple, non-invasive laser eye scan.
Alzheimer’s Disease has long been recognized as a growing diagnostic and therapeutic challenge – especially diagnosing the disease early when intervention could have a real impact. Unfortunately, current diagnostic methods are invasive, expensive, and complex, and are not yet widely available.
In this landscape, there is a significant unmet need for a highly accurate, safe, simple, non-invasive, and cost-effective test to aid in the early detection and diagnosis of Alzheimer’s disease.
To meet these needs, Cognoptix was co-founded by Dr. Lee Goldstein and Dr. Leo Chylack of the Brigham and Women’s Hospital in Boston. Together they developed an innovative system to detect beta amyloid proteins – a hallmark of Alzheimer’s disease – in the lens of the eye.
Pioneers in Alzheimer’s Diagnostics
Drs. Chylack and Goldstein and their research team discovered that they could detect beta amyloid aggregates in the lens of the eye and that those proteins could be measured and monitored with non-invasive tools, such as existing laser eye scanning technologies. Further research determined that suspect beta amyloid proteins could be detected in the eye prior to buildup of toxic plaques in the brains of genetically engineered Alzheimer’s mice. Cognoptix is currently developing combined optical scanning devices and diagnostic agents based on these discoveries for clinical, commercial and academic research use. The company is actively forming and seeking new strategic alliances with pharma partners and academic institutes, while preparing its diagnostic platform for clinical use.
technology
Cognoptix is developing technologies to enable non-invasive quantitative measurements of amyloid aggregates in the eye, to examine and measure deposits in specific areas of the lens as a means of early detection of Alzheimer’s disease.
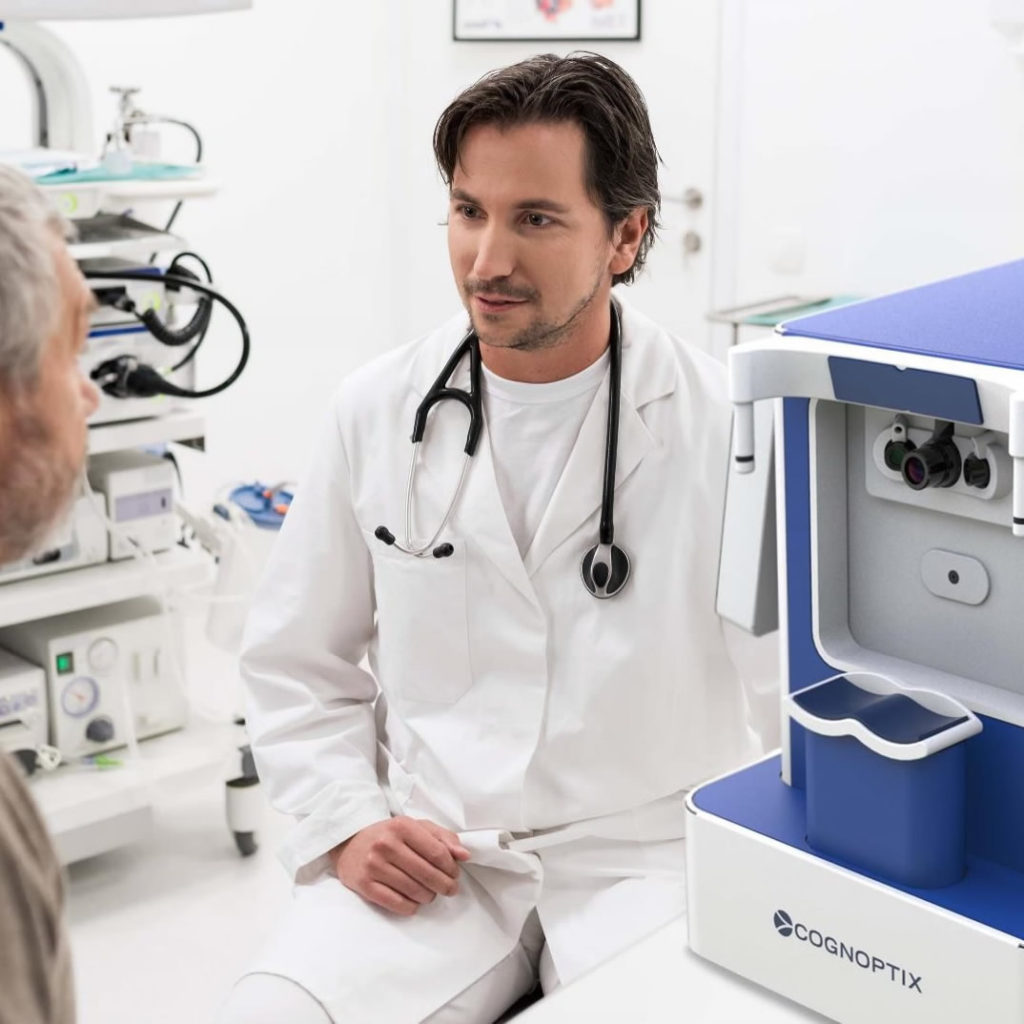
SAPPHIRE II System
SAPPHIRE Promises a New Hope
Cognoptix’s SAPPHIRE platform is a compact, easy to use, clinical device for in situ patient examination in a doctor’s office setting. The combined platform is currently available in the United States for investigational use only and is intended for clinical evaluation and measurement of beta-amyloid aggregation in anatomically defined regions of the anterior segment of the eye.
Comparable, Potentially Superior Performance Versus PET Scan
The results of a multicenter clinical trial of its SAPPHIRE II eye test, designed to identify Alzheimer’s Disease (AD) patients via a beta-amyloid signature in their eyes, has been published in the peer-reviewed Journal of Alzheimer’s Disease & Other Dementias (AJA).
By detecting a specific fluorescent signature of ligand-marked beta-amyloid in the supranucleus region of the human lens, SAPPHIRE II achieved a sensitivity of 85% and a specificity of 95% in differentiating 20 patients who were clinically diagnosed with probable AD from a group of age-matched, healthy volunteers. In addition, the SAPPHIRE II test showed excellent correlation to PET (positron emission tomography) amyloid brain imaging, the current gold standard for detecting beta-amyloid in the brain.
“The easy-to-use SAPPHIRE II eye scan has demonstrated the clinical potential to remake the paradigm for the way in which Alzheimer’s Disease is currently diagnosed and managed.”
Fluorescent Ligand Eye Scanning (FLES)
Fluorescent Ligand Eye Scanning (FLES) is a technique in which a compound composed of beta amyloid-specific small molecules is dropped into a patient’s eye, which is scanned by the SAPPHIRE instrument.
The small molecules are absorbed into the lens and bind to the amyloid aggregates. The FLES system excites the fluorescent ligands that bind to amyloid and quantitatively measures emissions in specific anatomical locations to biochemically confirm the presence of amyloid.
The binding compounds emit light in a specific, detectable range of wavelengths. If binding increases over time, a positive diagnosis can be made, enabling clinicians to track the progress of the disease in patients by measuring levels of fluorescence – as well as potentially enabling doctors and pharmaceutical researchers to monitor the efficacy of Alzheimer’s drugs in clinical trial settings.
management
SENIOR LEADERSHIP TEAM
CLINICAL ADVISORY BOARD
Jeffrey L. Cummings, MD, ScD
Jeffrey L. Cummings, MD, ScD, is a world-renowned Alzheimer’s researcher and leader of clinical trials. He is currently Director, Center for Neurodegeneration and Translational Neuroscience at the Cleveland Clinic, Lou Ruvo Center for Brain Health, Las Vegas, Nevada and Vice Chair for Research and Research Professor at the University of Nevada, Las Vegas, Department of Brain Health.
Dr. Cummings’ interests embrace clinical trials, developing new therapies for brain diseases, and the interface of neuroscience and society. He is the originator/author of the Neuropsychiatric Inventory (NPI), has authored or edited 43 books, and has published over 725 peer-reviewed papers.
Dr. Cummings has been recognized for his research and leadership contributions in the field of Alzheimer’s disease through prestigious lifetime achievement and other awards given by organizations such as: the national Alzheimer’s Association, the Society for Behavioral and Cognitive Neurology, the American Association of Geriatric Psychiatry, the International Society of CNS Drug Development, and the American Geriatrics society.
Dr. Cummings graduated magna cum laude from the University of Wyoming, Laramie, obtained his MD degree from the University of Washington, Seattle, and completed his Neurology residency and a Fellowship in Behavioral Neurology at Boston University, Boston, Massachusetts. This training was followed by a Research Fellowship in Neuropathology and Neuropsychiatry at the National Hospital for Nervous Diseases, Queen Square, London, England.
Jeffrey L. Cummings, MD, ScD
Jeffrey L. Cummings, MD, ScD, is a world-renowned Alzheimer’s researcher...
Lee Goldstein, MD, PhD
Dr. Goldstein is Associate Professor of Psychiatry, Radiology, Neurology, Pathology & Laboratory Medicine, Biomedical Engineering, and Electrical & Computer Engineering at Boston University School of Medicine, College of Engineering, and Photonics Center. He leads multidisciplinary research teams as Director of the Molecular Aging & Development Laboratory, Center for Biometallomics, and Neurotrauma Laboratory at Boston University School of Medicine. He co-directs the Biomarker Core, NIH-funded Boston University Alzheimer’s Disease Center, and the newly established Center for Translational Neuroimaging. While at Harvard Medical School, Dr. Goldstein discovered Alzheimer’s disease β-amyloid (Aβ) pathology in the lens of the eye (Goldstein et al., Lancet, 2003), the first evidence of Alzheimer’s pathology outside the brain. He predicted and subsequently identified Aβ lens pathology as the origin of cataracts in Down syndrome (Trisomy 21), a chromosomal disorder in which early-onset β-amyloid brain pathology is an invariant feature.
In 2001, Dr. Goldstein co-founded Neuroptix (now Cognoptix) with the goal of developing a drug-device combination eye scanner for early detection and monitoring of Alzheimer’s disease. He served the company for over a decade and recently rejoined Cognoptix to help move the Sapphire II system to the forefront of AD diagnostic technologies. He received his baccalaureate from Columbia, MD-PhD degrees from Yale, and clinical and research fellowships at Massachusetts General Hospital, Harvard Medical School, where he joined the faculty. In 2008, he moved to Boston University. His research focuses on Alzheimer’s disease, lens biology, traumatic brain injury, chronic traumatic encephalopathy, and laser-based diagnostic technologies.
Lee Goldstein, MD, PhD
Dr. Goldstein is Associate Professor of Psychiatry, Radiology, Neurology, Pathology...
Marwan Noel Sabbagh, MD, FAAN
Marwan Noel Sabbagh, MD, board certified neurologist and geriatric neurologist, hopes to work himself out of a job. Considered one of the leading experts in Alzheimer’s and dementia, he is the Camille and Larry Ruvo Endowed Chair for Brain Health and Director of Translational Research at Cleveland Clinic Lou Ruvo Center for Brain Health in Las Vegas. Dr Sabbagh has dedicated his career to finding a cure for Alzheimer’s and other age-related neurodegenerative diseases.
Dr. Sabbagh is a leading investigator for many prominent national Alzheimer’s prevention and treatment trials. Dr. Sabbagh is on the editorial board for Journal of Alzheimer’s Disease and BMC Neurology. He is now editor in chief of Neurology and Therapy. He has authored and co-authored almost 370 medical and scientific articles on Alzheimer’s research.
Dr. Sabbagh is the author of The Alzheimer’s Answer: Reduce Your Risk and Keep Your Brain Healthy, with foreword by Justice Sandra Day O’Connor, and of The Alzheimer’s Prevention Cookbook: 100 Recipes to Boost Brain Health. He has edited Palliative Care for Advanced Alzheimer’s and Dementia: Guidelines and Standards for Evidence Based Care and Geriatric Neurology, published in 2014 and Fighting for my Life: living in the shadow of Alzheimer’s disease in 2019.
He has been recognized with numerous awards, including WestMarc Innovator Award, 2015; Fellow of the American Academy of Neurology, 2004.
Dr. Sabbagh earned his undergraduate degree from the University of California, Berkeley and his medical degree from the University of Arizona in Tucson. He received his residency training in neurology at Baylor College of Medicine, Houston, Texas, and completed his fellowship in geriatric neurology and dementia at the University of California, San Diego School of Medicine, where he served on the faculty as assistant professor. Before joining the faculty of the Cleveland Clinic, he was at the Barrow Neurological Institute where he served for three years, and prior to that, he was the director of the Banner Sun Health Research Institute for 15 years.
Marwan Noel Sabbagh, MD, FAAN
Marwan Noel Sabbagh, MD, board certified neurologist and geriatric neurologist,...
Dr. Michael D. Devous, Sr.
Dr. Devous recently retired as Vice President of Imaging Development at Avid Radiopharmaceuticals, a wholly owned subsidiary of Eli Lilly and Company. His responsibilities included serving on the Executive Committee, directing the Imaging Sciences Department, and designing, executing, analyzing, and interpreting clinical trials. He also assisted in development and implementation of online and in-person training for the use of imaging biomarkers. He also served on clinical trial design, analysis, and interpretation teams for therapeutics developed by Eli Lilly and Company with a focus on imaging and non-imaging (e.g., plasma) biomarkers and their role in therapeutic clinical trials. He was also previously Professor of Radiology, Neurology, and Bioengineering, Director of the Alzheimer’s Disease Center Neuroimaging Core, Co-Principal Investigator of the North Texas Traumatic Brain Injury Model System, and Associate Director of the Nuclear Medicine Center at the University of Texas Southwestern Medical Center, Dallas, Texas. He remains an Adjunct Professor of Neurology at UT Southwestern and of Behavioral and Brain Sciences at the University of Texas at Dallas. He has authored more than 200 peer-reviewed articles, 375 abstracts, and 30 books or book chapters. Dr. Devous is past president of the Society of Nuclear Medicine, the Education and Research Foundation, and the Brain Imaging Council. He is a former chair of the Food and Drug Administration Medical Imaging Drugs Advisory Committee. He has received numerous awards, including: Presidential Distinguished Service Award – Society of Nuclear Medicine; Scientific Merit First Award – American Speech, Language and Hearing Association; and the Kuhl-Lassen Award, the highest award of the Brain Imaging Council and the Society of Nuclear Medicine.
His research focuses on advancing understanding of the pathobiology of neurologic and psychiatric disorders, and normal brain function, through molecular, functional, and structural neuroimaging techniques.
Dr. Michael D. Devous, Sr.
Dr. Devous recently retired as Vice President of Imaging Development...
Martin R. Farlow, MD
Martin R. Farlow, is Professor Emeritus in the Department of Neurology at Indiana University and co-director of the Alzheimer’s Disease Center at Indiana University.
Dr. Farlow is one of the country’s leaders in Alzheimer disease (AD) and other cognitive disorders. An expert clinical trialist, Dr. Farlow has conducted over 150 clinical studies of new drug candidates for the treatment of AD and related dementias.
Dr Farlow also has clinically characterized and helped determine genetic linkage for several familial dementias including the second mutation associated with autosomal dominantly inherited Alzheimer’s disease, Gerstmann-Straussler-Scheinker disease, and Multi Systems Tauopathy with Dementia
He has lectured on the topics of aging, dementia, and AD at more than 300 meetings, conferences, and hospitals/medical schools throughout the world. A prolific author, Dr. Farlow has presented more than 467 abstracts at professional meetings and has authored or co-authored more than 455 articles published in peer-reviewed journals.
Dr. Farlow is a member of many professional associations including the American Academy of Neurology and the American Neurological Association. He is also a founding member of both the American Society of Experimental Neurotherapeutics and the International Society for CNS Clinical Trials and Methodology
Dr. Farlow received his medical degree from Indiana University School of Medicine. He completed an internship in Internal Medicine and a residency in neurology at the Indiana University Hospitals.
Martin R. Farlow, MD
Martin R. Farlow, is Professor Emeritus in the Department of...
BOARD OF DIRECTORS
Lee Goldstein, MD, PhD
Dr. Goldstein is Associate Professor of Psychiatry, Radiology, Neurology, Pathology & Laboratory Medicine, Biomedical Engineering, and Electrical & Computer Engineering at Boston University School of Medicine, College of Engineering, and Photonics Center. He leads multidisciplinary research teams as Director of the Molecular Aging & Development Laboratory, Center for Biometallomics, and Neurotrauma Laboratory at Boston University School of Medicine. He co-directs the Biomarker Core, NIH-funded Boston University Alzheimer’s Disease Center, and the newly established Center for Translational Neuroimaging. While at Harvard Medical School, Dr. Goldstein discovered Alzheimer’s disease β-amyloid (Aβ) pathology in the lens of the eye (Goldstein et al., Lancet, 2003), the first evidence of Alzheimer’s pathology outside the brain. He predicted and subsequently identified Aβ lens pathology as the origin of cataracts in Down syndrome (Trisomy 21), a chromosomal disorder in which early-onset β-amyloid brain pathology is an invariant feature.
In 2001, Dr. Goldstein co-founded Neuroptix (now Cognoptix) with the goal of developing a drug-device combination eye scanner for early detection and monitoring of Alzheimer’s disease. He served the company for over a decade and recently rejoined Cognoptix to help move the Sapphire II system to the forefront of AD diagnostic technologies. He received his baccalaureate from Columbia, MD-PhD degrees from Yale, and clinical and research fellowships at Massachusetts General Hospital, Harvard Medical School, where he joined the faculty. In 2008, he moved to Boston University. His research focuses on Alzheimer’s disease, lens biology, traumatic brain injury, chronic traumatic encephalopathy, and laser-based diagnostic technologies.
Lee Goldstein, MD, PhD
Dr. Goldstein is Associate Professor of Psychiatry, Radiology, Neurology, Pathology...
R. Gregg Stone
R. Gregg Stone has been active in the investment business for forty years from the seed stages of companies’ development as an angel investor and board member to selecting public market managers. Most of his investments have been related to the delivery of healthcare. He is an active member of Launchpad Venture Group, the largest angel group in the Northeast. Gregg currently serves as a director of Cambridge Trust Company and a private company. Until its sale to eRT in 2015, he served as the Chairman of PHT Corp., a pharmaceutical service company based in Charlestown, Massachusetts. Prior and current investments include NovaCare, Health Dialog, CardioFocus and Iora Healthcare. Before founding Kestrel in 1996, Gregg worked at Pell, Rudman & Co. Inc. (now Atlantic Trust Company), a Boston-based investment adviser, after practicing law with Hemenway & Barnes in Boston. He holds J.D. and A.B. degrees from Harvard. He is a director of the Boys & Girls Clubs of Boston and Camp Pasquaney and a trustee emeritus of the Beth Israel Deaconess Medical Center. He is a Corporate Trustee of The Trustees of Reservations, and Co-chair of the Overseers of Boston Summer Search. Gregg coached his daughter, Gevvie Stone, who won a silver medal in the women’s single scull at the Rio Olympics.
R. Gregg Stone
R. Gregg Stone has been active in the investment business...
John Conley
John Conley co-founded Gilliam Capital LLC, a life science investment firm, and is a Member and Director of Launchpad Venture Group in Boston. He is a board member for three healthcare startups and works with a variety of social enterprises. He has been the Chief Operating Officer of Entrepreneurship for All, a nonprofit that is accelerating economic and social impact through fostering entrepreneurship in mid-sized cities.
He was a co-founder of the RNA interference therapeutics company Alnylam Pharmaceuticals where he held the position of Vice President, Strategy and Finance and CFO through to its successful 2004 IPO. He was Vice President of Business Development at Transkaryotic Therapies through to its acquisition by Shire Pharmaceuticals. Over ten years at Biogen he served in several marketing, business development, sales and finance positions, including Country Manager – United Kingdom and Ireland, and Treasurer. He was a Manager at the strategy-consulting firm of Bain & Company.
Mr. Conley graduated with a BS in Economics from the University of Pennsylvania’s Wharton School and an MBA from the Yale School of Management. He was a 2014 Fellow in the Advanced Leadership Initiative at Harvard University.
John Conley
John Conley co-founded Gilliam Capital LLC, a life science investment...
news
COMPANY NEWS
Dr. Marwan Sabbagh Joins the Cognoptix Scientific Advisory Board
Cognoptix announced that Marwan Noel Sabbagh, MD has joined Cognoptix’ Scientific Advisory Board. Dr. Sabbagh will play a pivotal role in helping guide the strategic direction of the company to support the continued efforts of achieving FDA approval for the Sapphire II system.
A pioneer in creating and developing a simple, innovative, non-invasive eye scanning test to aid in the early detection and diagnosis of Alzheimer’s Disease: Cognoptix
Various healthcare researchers and companies are trying to innovate new methods to diagnose Alzheimer’s, but Cognoptix stands out from the rest with its Sapphire II system, detecting Alzheimer’s disease through a simple scan of the lens of the eye.
Cognoptix Appoints New Chief Medical Officer, Dr. Carl Sadowsky, MD, and Chief Financial Officer, Mike Kaswan, MBA, to Management Team
Cognoptix announced that Carl H. Sadowsky, MD, will serve as the company’s new Chief Medical Officer (CMO), and Mike Kaswan, MBA, will be Cognoptix’s Chief Financial Officer (CFO).
Cognoptix Announces Formation of a New Board of Directors
As another milestone of its Series R restart, recapitalization, and revitalization, the company named a new Board of Directors under President and CEO Susanne Wilke, Ph.D.
Cognoptix Co-Founder Lee Goldstein, M.D., Ph.D. Joins Cognoptix Board of Directors and Scientific Advisory Board
Cognoptix announces its co-founder, Lee Goldstein, M.D., Ph.D. has joined Cognoptix’s Scientific Advisory Board, serving as Co-Chair, and the company’s Board of Directors.
Cognoptix Announces New Scientific Advisory Board with Alzheimer’s Research Leaders
Cognoptix announced the formation of its new Scientific Advisory Board (SAB) with appointment of five world-class scientists and clinical researchers in AD.
Cognoptix Revitalizes, Appoints New Chief Executive Officer Dr. Susanne Wilke, PhD, Raises Oversubscribed $2.6m Series R
Cognoptix announces the closing of an over-subscribed Series R financing round.
Cognoptix developing an Alzheimer’s disease test using the eye
Cognoptix Inc. CEO Susanne Wilke sat down with Proactive’s Christine Corrado at the Biotech Showcase 2020 in San Francisco. The privately held, Concord, Massachusetts-based company develops non-invasive eye tests for the early diagnosis of Alzheimer’s disease.
Cognoptix to Present at Biotech Showcase™ 2020, San Francisco, CA
Cognoptix has scheduled a company presentation at Biotech Showcase 2020, to be held January 13–15, during the most important week in healthcare at the Hilton San Francisco Union Square.
Cognoptix Investor Presentation
September 2020
“Cognoptix: Pioneering the Early Detection of Alzheimer’s Disease”
Cognoptix was recently profiled by MedTech Outlook as one of the Top 10 Non-Invasive Device Solution Providers for 2020.
“A pioneer in creating and developing a simple, innovative, non-invasive eye scanning test to aid in the early detection and diagnosis of Alzheimer’s Disease: Cognoptix”
Various healthcare researchers and companies are trying to innovate new methods to diagnose Alzheimer’s, but Cognoptix stands out from the rest with its Sapphire II system, detecting Alzheimer’s disease through a simple scan of the lens of the eye.
INDUSTRY NEWS

FDA grants historic approval to Alzheimer’s drug designed to slow cognitive decline
The Food and Drug Administration approved the first new treatment for Alzheimer’s disease in nearly two decades, a landmark decision that has been eagerly awaited by millions of Americans
FDA Approves Aduhelm, First Targeted Alzheimer’s Therapy
For the first time in almost 18 years, the U.S. Food and Drug Administration (FDA) has approved a new treatment — Aduhelm, also known as aducanumab — for Alzheimer’s disease, and a first targeted treatment for patients.

Lecanemab Safely, Quickly Lowers Amyloid Clumps in Trial
Eisai and Biogen’s experimental therapy lecanemab (BAN2401) safely leads to rapid and sustained reductions in the brain levels of beta-amyloid — the protein that forms toxic clumps in Alzheimer’s — in people in early disease stages, according to one-year, preliminary data from a Phase 2 trial’s ongoing extension phase.

The Battle Over an Alzheimer’s Treatment
Biogen’s promising drug is caught in the FDA’s political and bureaucratic limbo.

Eli Lilly Offers More Positive Data on Its Alzheimer’s Drug
Eli Lilly is presenting data Saturday that bolsters its claims that its experimental Alzheimer’s antibody therapy slows down the disease in early-stage patients

The Other Pandemic: What to Do About the Coming Alzheimer’s Crisis
With the world in the midst of an aging boom, the number of people living with Alzheimer’s or some form of dementia is expected to triple by 2050 to 152 million—a bit more than the population of Russia today.

Eli Lilly adds $23B in market cap as investors rush on promise that this will be the amyloid Alzheimer’s drug that works
While the world awaits the FDA’s decision on Biogen’s controversial aducanumab pitch, a Big Pharma rival says they now have data to support their own amyloid-clearing Alzheimer’s drug.

Lilly Alzheimer’s Drug Helped Patients in Small Trial
An experimental Alzheimer’s drug from Eli Lilly & Co. helped patients in a small trial, the company said, renewing hopes that researchers are closing in on therapies that can fight the disease.

New Film “The Artist’s Wife” Tells the Story of a Family Navigating an Alzheimer’s Diagnosis
The name “Dolby” is synonymous with the power of sound thanks to American inventor Ray Dolby, who passed away in September 2013 from Alzheimer’s disease.

Super-Agers Show Resistance to Tau and Amyloid Accumulation
PET neuroimaging reveals super-agers and those whose cognitive skills are above the norm for an advanced age have an increased resistance to tau and amyloid proteins.
contact us
COGNOPTIX, INC.
33 Boston Post Road West
Suite 140
Marlborough, MA 01752
(508) 251-1546










